Categories
- FQA (108)
 Quantum computing principles have evolved far beyond theoretical physics laboratories. Today, practical quantum devices serve millions of patients worldwide through non-invasive health analysis systems. These sophisticated instruments combine quantum mechanics with modern diagnostic technology to deliver comprehensive health assessments in minutes.This comprehensive guide explores the international safety standards governing quantum devices, helping Nigerian business professionals understand certification requirements, compliance frameworks, and market entry strategies. Whether you operate a medical equipment distribution company, manage health clinics, or seek profitable business opportunities in Nigeria’s growing wellness sector, this article provides essential knowledge for success. Discover how MAIKONG maintains rigorous international safety standards while delivering innovative health analysis solutions to African markets.
Quantum computing principles have evolved far beyond theoretical physics laboratories. Today, practical quantum devices serve millions of patients worldwide through non-invasive health analysis systems. These sophisticated instruments combine quantum mechanics with modern diagnostic technology to deliver comprehensive health assessments in minutes.This comprehensive guide explores the international safety standards governing quantum devices, helping Nigerian business professionals understand certification requirements, compliance frameworks, and market entry strategies. Whether you operate a medical equipment distribution company, manage health clinics, or seek profitable business opportunities in Nigeria’s growing wellness sector, this article provides essential knowledge for success. Discover how MAIKONG maintains rigorous international safety standards while delivering innovative health analysis solutions to African markets.Understanding what constitutes a quantum device requires examining both theoretical foundations and practical applications. In contemporary healthcare markets, these sophisticated instruments leverage principles from quantum physics and quantum mechanics to analyze biological systems at cellular and molecular levels.
Quantum computing emerged from theoretical physics research conducted throughout the twentieth century. Scientists discovered that particles at subatomic scales behave according to quantum mechanical principles fundamentally different from classical physics laws governing everyday objects. These discoveries revolutionized multiple industries, including healthcare diagnostics.
Modern quantum devices designed for health analysis measure electromagnetic frequencies emitted by human cells. Every biological system in the human body generates unique electromagnetic signatures. Quantum computing algorithms process these signals through advanced mathematical models, producing detailed health reports within minutes.
The transition from theoretical quantum mechanics to practical quantum device applications involved decades of research. Early quantum computers required massive laboratory infrastructure with temperatures near absolute zero. Today’s health analysis quantum devices operate at room temperature using sophisticated sensors that detect minute electromagnetic variations.

Conventional computers process information using binary bits representing either zero or one. Quantum computers and quantum devices utilize qubits that can exist in multiple states simultaneously through a phenomenon called superposition. This fundamental difference enables quantum systems to analyze complex biological data patterns impossible for classical computers to process efficiently.
In healthcare applications, this distinction becomes critically important. Conventional computers would require hours to analyze the electromagnetic signatures from multiple organ systems. Quantum device technology completes comprehensive body scans in approximately ninety seconds, generating reports across fifty-four health parameters.
The power quantum technology brings to medical diagnostics extends beyond speed. Quantum algorithms identify subtle patterns in electromagnetic data that traditional diagnostic methods might miss. This capability makes quantum devices valuable tools for preventive healthcare and early detection of potential health issues.
The healthcare industry utilizes several categories of quantum devices, each serving specific diagnostic purposes. Understanding these categories helps distributors and healthcare providers select appropriate equipment for their target markets and patient populations.
Quantum resonance analyzers represent the most widely adopted category in Nigerian and African healthcare markets. These non-invasive quantum devices assess health status through electromagnetic frequency analysis. Patients simply hold a sensor while the quantum device collects and analyzes electromagnetic signals from their body.
Research-grade quantum computers exist primarily in advanced laboratory settings. These sophisticated quantum systems contribute to drug discovery, genetic research, and complex medical modeling. While not directly accessible to typical healthcare providers, their research outputs influence the quantum algorithms embedded in commercial quantum devices.

Global safety standards protect patients, healthcare providers, and end-users of medical quantum devices. Multiple international organizations establish comprehensive frameworks governing device manufacturing, testing, certification, and market distribution. Nigerian entrepreneurs entering the quantum device market must understand these frameworks to ensure compliance and build customer trust.
The International Organization for Standardization maintains comprehensive standards for medical quantum devices through its ISO 13485 certification framework. This standard specifies requirements for quality management systems where organizations need to demonstrate their ability to provide medical devices meeting customer and regulatory requirements.
ISO 13485 certification involves rigorous auditing of manufacturing processes, quality control procedures, and risk management systems. Manufacturers of quantum devices must document every aspect of their production process, from component sourcing through final product testing. Regular surveillance audits ensure ongoing compliance with international standards.
For Nigerian distributors, partnering with ISO-certified manufacturers provides significant competitive advantages. Hospitals, clinics, and wellness centers increasingly require certification documentation before purchasing medical equipment. MAIKONG maintains comprehensive ISO certification across its quantum device product line, providing distributors with complete documentation packages supporting market entry.
| ISO Standard | Quantum Device Application | Certification Scope | Renewal Period |
| ISO 13485:2016 | Medical device quality management systems | Manufacturing, design, installation, servicing | Annual surveillance, three-year recertification |
| ISO 14971:2019 | Medical device risk management | Risk analysis, evaluation, control, monitoring | Ongoing process review requirements |
| ISO 10993 Series | Biological evaluation of medical devices | Biocompatibility testing for patient contact components | Product-specific testing protocols |
| ISO 80601-2-56 | Clinical thermometers with patient contact | Safety and performance requirements | Version-specific compliance verification |
| ISO 15223-1 | Medical device symbols and labeling | User interface, packaging, documentation symbols | Updated with symbol standard revisions |
The CE marking system enables quantum device manufacturers to demonstrate conformity with European Economic Area health, safety, and environmental protection standards. This marking serves as a mandatory requirement for medical device sales throughout European markets and provides credibility in international markets including Nigeria.
Obtaining CE certification for quantum devices requires conformity assessment procedures specific to device classification. Most health analysis quantum devices fall under Class IIa medical device categories, requiring technical documentation review and quality system assessment by notified bodies.
The certification process examines multiple aspects of quantum device performance and safety. Manufacturers must provide comprehensive technical files documenting device design, manufacturing processes, clinical evaluation reports, and post-market surveillance plans. Electromagnetic compatibility testing ensures quantum devices neither interfere with other medical equipment nor experience interference affecting their operation.

FDA classification systems categorize medical quantum devices based on risk levels and intended use. Class I devices present minimal risk and face general controls. Class II devices require special controls including performance standards and post-market surveillance. Class III devices present the highest risk and demand pre-market approval through clinical trials.
Most diagnostic quantum devices utilizing electromagnetic frequency analysis receive Class II classification under FDA frameworks. Manufacturers must submit 510(k) pre-market notifications demonstrating substantial equivalence to legally marketed devices. This process includes performance testing data, biocompatibility studies, and electromagnetic compatibility verification.
Nigerian distributors benefit from FDA recognition even when operating exclusively in African markets. Many Nigerian healthcare institutions follow international best practices, preferring equipment meeting multiple international standards. MAIKONG’s commitment to comprehensive international compliance positions distributors for success across diverse market segments.
MAIKONG Quantum Health Analyzer systems meet comprehensive international safety requirements including ISO 13485, CE marking, and industry-leading quality standards. Access complete certification documentation and technical specifications designed specifically for African market requirements.
Different geographical regions maintain unique regulatory frameworks governing quantum device importation, distribution, and use. Understanding these regional variations enables Nigerian distributors to navigate compliance requirements efficiently while expanding their market reach across West African Economic Community nations.
The African Union developed harmonized medical device regulations promoting consistent safety standards across member nations. These frameworks facilitate trade between African countries while protecting public health through evidence-based regulatory approaches adapted to African healthcare contexts.
Nigeria actively participates in African Union harmonization initiatives through the West African Health Organization. This participation influences national regulatory requirements for quantum devices and other medical equipment. Distributors benefit from understanding both Nigerian-specific regulations and broader regional frameworks.
Harmonization efforts emphasize post-market surveillance and vigilance systems. Distributors must establish mechanisms for reporting adverse events, device malfunctions, and customer feedback. These systems protect end-users while helping manufacturers improve quantum device performance based on real-world usage data.
NAFDAC maintains regulatory authority over medical devices including quantum devices entering Nigerian markets. The agency requires product registration before commercial distribution, involving documentation review, facility inspection, and ongoing compliance monitoring.
Registration processes vary based on device classification and manufacturer location. International manufacturers with established safety certifications from recognized authorities benefit from streamlined registration procedures. MAIKONG’s comprehensive international certifications facilitate efficient NAFDAC registration for Nigerian distributors.
Distributors must maintain detailed records of quantum device sales, including purchaser information, installation locations, and service histories. NAFDAC conducts periodic market surveillance activities verifying compliance with registration requirements and quality standards. Partnering with manufacturers providing comprehensive support documentation ensures smooth regulatory interactions.

ECOWAS member states increasingly coordinate medical device regulations through regional standardization initiatives. These efforts reduce barriers to cross-border trade while maintaining consistent safety standards protecting West African populations.
For Nigerian distributors, regional harmonization creates opportunities for market expansion beyond national borders. Quantum devices registered through Nigerian regulatory pathways may qualify for simplified registration procedures in neighboring countries. This regional approach enables distributor growth strategies leveraging economies of scale.
Technical standards developed through ECOWAS frameworks often align with international ISO standards and European CE marking requirements. This alignment benefits distributors partnering with globally certified manufacturers like MAIKONG, whose quantum devices meet or exceed regional technical specifications.
Electrical safety represents a critical component of quantum device certification frameworks. These sophisticated instruments combine sensitive electronic components with patient-contact interfaces, requiring comprehensive safety testing and ongoing quality verification.
The International Electrotechnical Commission establishes safety requirements for medical electrical equipment through its IEC 60601 standard series. This comprehensive framework addresses potential electrical hazards including electric shock, mechanical hazards, excessive temperatures, and electromagnetic interference.
Quantum devices must comply with IEC 60601-1 general requirements plus applicable particular standards. Testing protocols verify proper insulation, protective earth connections, leakage current limits, and electromagnetic compatibility. Equipment must function safely under normal conditions and reasonably foreseeable fault conditions.
Nigerian distributors should verify manufacturers provide complete IEC 60601 test reports with their quantum devices. These reports document compliance with specific safety parameters relevant to device classification and intended use. MAIKONG maintains comprehensive electrical safety documentation supporting distributor compliance obligations.
Electromagnetic compatibility ensures quantum devices operate correctly within typical electromagnetic environments while not generating excessive electromagnetic interference affecting other equipment. These requirements become particularly important in clinical settings where multiple medical devices operate simultaneously.
Testing protocols examine both emissions and immunity characteristics. Emission testing verifies quantum devices do not generate electromagnetic interference exceeding specified limits. Immunity testing confirms devices continue functioning correctly when exposed to electromagnetic disturbances from other equipment, mobile phones, and radio transmitters.
Nigerian healthcare facilities increasingly implement multiple technology systems within limited physical spaces. Electromagnetic compatibility certification ensures quantum devices integrate seamlessly into existing clinical infrastructure without disrupting other critical medical equipment operations.
Global markets operate on various electrical standards with different voltage levels and frequency specifications. Nigerian electrical systems typically provide 230 volts at 50 hertz frequency. Quantum devices distributed in Nigerian markets must accommodate these specifications or include appropriate voltage conversion equipment.
MAIKONG quantum devices incorporate universal power supplies supporting voltage ranges from 110 to 240 volts and frequencies of 50 to 60 hertz. This flexibility enables operation across diverse markets without requiring separate models or external voltage converters. Distributors benefit from simplified inventory management and broader application possibilities.
Power supply stability significantly impacts quantum device performance. Built-in protection circuits guard against voltage fluctuations, power surges, and electrical transients common in developing market electrical infrastructure. These protective features extend equipment lifespan while ensuring consistent diagnostic accuracy regardless of local power quality variations.
| Safety Parameter | IEC 60601 Requirement | Test Method | MAIKONG Compliance |
| Earth leakage current | Maximum 500 microamperes normal condition | Direct measurement with calibrated instruments | Typical 150 microamperes, well below limit |
| Patient leakage current | Maximum 100 microamperes for BF type | Applied part to earth measurement protocol | Certified at 45 microamperes average |
| Insulation resistance | Minimum 10 megohms at 500V DC test | High voltage insulation testing equipment | Exceeds 50 megohms consistently |
| Electromagnetic emissions | CISPR 11 Group 1 Class B limits | Radiated and conducted emission measurement | Full compliance verified annually |
| Electromagnetic immunity | IEC 61000-4 series immunity levels | Controlled electromagnetic field exposure | Passes all immunity test protocols |
Modern quantum devices incorporate sophisticated software managing data collection, analysis algorithms, and results presentation. Software safety requirements ensure these systems function reliably, protect patient data, and maintain diagnostic accuracy throughout their operational lifetime.
IEC 62304 establishes software lifecycle processes for medical device software development. This standard addresses software architecture, design, implementation, verification, and maintenance activities. Manufacturers must demonstrate systematic approaches to software development reducing risks of software failures affecting patient safety or diagnostic accuracy.
Quantum algorithms embedded within health analysis quantum devices require rigorous validation protocols. Software verification testing confirms algorithms produce accurate, reproducible results across diverse patient populations. Validation studies demonstrate clinical relevance of analysis outputs supporting healthcare decision-making.
MAIKONG maintains comprehensive software documentation supporting regulatory compliance and distributor technical support requirements. Software packages include detailed user manuals, technical specifications, and installation guides enabling efficient deployment across Nigerian healthcare facilities.

Quantum device software processes sensitive patient health information requiring robust data protection measures. International frameworks including General Data Protection Regulation principles influence medical device data handling requirements even in markets outside European jurisdictions.
Nigerian Data Protection Regulation establishes requirements for processing personal information including health data. Quantum device software must implement appropriate technical and organizational measures protecting patient information from unauthorized access, disclosure, or loss. Encryption, access controls, and audit logging represent essential security features.
MAIKONG software incorporates multilayer data protection architecture. Patient information remains stored locally on healthcare provider systems rather than transmitting to external servers. This approach gives Nigerian healthcare facilities complete control over patient data while simplifying compliance with data protection regulations.
Quantum device software must operate reliably across various computing platforms prevalent in target markets. MAIKONG Quantum Analyzer software supports Windows XP, Windows 7, Windows Vista, Windows 8, Windows 10, and Windows 11 operating systems. This broad compatibility enables deployment in healthcare facilities with diverse existing computer infrastructure.
The software does not support Android, Apple macOS, Linux, or Unix operating systems. Nigerian distributors should clearly communicate these requirements to potential customers during sales processes. Most Nigerian healthcare facilities operate Windows-based computer systems, making compatibility concerns minimal in typical deployment scenarios.
System requirements remain modest, enabling quantum device software operation on standard office computers without requiring specialized hardware investments. Minimum specifications include 2GB RAM, 500MB available storage space, and standard USB ports for device connectivity. These accessible requirements reduce total ownership costs for healthcare providers.
Access comprehensive MAIKONG Quantum Analyzer software packages, user manuals, and technical specifications. Nigerian distributors receive complete documentation supporting customer training, technical support, and regulatory compliance requirements.
Regulatory frameworks require clinical evidence demonstrating quantum device safety and performance claims. Clinical validation studies establish diagnostic accuracy, reproducibility, and clinical utility supporting device marketing claims and healthcare provider adoption.
Medical Device Regulation frameworks require manufacturers to compile clinical evaluation reports summarizing available clinical data supporting device safety and performance. These reports analyze clinical investigations, scientific literature, and post-market clinical follow-up data demonstrating devices achieve intended purposes without unacceptable risks.
Clinical evaluation represents an ongoing process throughout quantum device lifecycle. Manufacturers must continuously monitor emerging clinical data, adverse event reports, and scientific publications relevant to their devices. Post-market surveillance activities generate real-world evidence supplementing pre-market clinical studies.
MAIKONG maintains comprehensive clinical documentation supporting quantum resonance analyzer performance claims. Studies demonstrate high correlation between device analysis outputs and conventional diagnostic methods across multiple health parameters. Nigerian distributors receive clinical summaries supporting customer education and professional credibility.
Quantum device accuracy validation involves comparing device outputs against established reference methods. For health analysis quantum devices, validation studies examine correlations between electromagnetic frequency measurements and clinical laboratory test results, physical examinations, and patient health outcomes.
Reproducibility testing confirms quantum devices generate consistent results when measuring the same subject repeatedly under controlled conditions. Variability in measurement results must remain within acceptable limits defined by device intended use and clinical significance of measured parameters.
Environmental condition testing verifies quantum device performance across temperature ranges, humidity levels, and altitude variations encountered in typical use environments. Nigerian distributors benefit from devices tested under tropical climate conditions similar to West African environments, ensuring reliable performance regardless of local environmental factors.

Post-market surveillance systems track adverse events, device malfunctions, and near-miss incidents throughout quantum device operational life. Regulatory authorities require manufacturers and distributors to establish vigilance systems capturing, investigating, and reporting safety concerns.
Nigerian distributors must implement processes for receiving and documenting customer complaints, device malfunctions, and potential safety issues. Serious incidents require reporting to NAFDAC following established timeframes and documentation requirements. MAIKONG provides distributor support for incident investigation and regulatory reporting obligations.
Trend analysis of post-market data identifies potential safety or performance issues requiring corrective actions. Manufacturers may issue safety notices, implement software updates, or initiate device recalls when monitoring reveals concerns affecting patient safety or diagnostic accuracy. Proactive vigilance protects patients while demonstrating distributor commitment to quality and safety.
Comprehensive quality management systems ensure consistent quantum device manufacturing processes producing products meeting specified requirements. ISO 13485 certification demonstrates manufacturer commitment to quality throughout design, production, distribution, and servicing activities.
Quality systems establish formal design and development processes for quantum devices. These processes include design planning, input specification, output verification, design review, validation, and transfer to manufacturing. Documentation demonstrates systematic approaches reducing risks of design flaws affecting safety or performance.
Risk management activities integrate throughout design and development processes. Manufacturers identify potential hazards, estimate risks, implement risk controls, and verify control effectiveness. This systematic risk management produces quantum devices with acceptable risk-benefit profiles supporting regulatory approval and market acceptance.
Design validation confirms quantum devices meet user needs and intended uses under actual or simulated use conditions. Clinical evaluations, usability studies, and performance testing provide objective evidence supporting design validation conclusions. MAIKONG conducts comprehensive validation studies ensuring devices perform reliably in diverse healthcare settings.
Quality systems establish controls ensuring consistent quantum device manufacturing. Process validation studies demonstrate manufacturing processes reliably produce devices meeting specifications. In-process controls monitor critical process parameters preventing defects during production.
Component and material controls ensure incoming materials meet specified requirements. Supplier qualification processes verify component suppliers maintain appropriate quality systems. Incoming inspection activities confirm materials comply with purchase specifications before entering production processes.
Environmental controls maintain appropriate manufacturing conditions for quantum device production. Temperature, humidity, cleanliness, and electrostatic discharge protection levels align with device requirements. Environmental monitoring demonstrates ongoing compliance with established conditions supporting product quality.

Comprehensive testing protocols verify quantum devices meet specifications before market release. Testing includes electrical safety verification, electromagnetic compatibility assessment, software validation, performance testing, and packaging integrity evaluation. Test records provide objective evidence of device quality and compliance.
Final inspection processes examine completed quantum devices confirming they meet all requirements. Visual inspection identifies cosmetic defects. Functional testing verifies operational performance. Documentation review confirms all required records accompany finished devices. Only devices passing all inspection criteria receive release for distribution.
Calibration programs ensure measuring and test equipment maintains accuracy throughout its operational life. Calibration procedures trace measurements to national or international standards providing confidence in test results. Regular calibration intervals prevent measurement drift affecting product quality assessments.
MAIKONG implements industry-leading quality management systems exceeding basic certification requirements. Every quantum device undergoes rigorous testing protocols validating performance specifications and safety standards.
Nigerian distributors benefit from comprehensive quality documentation supporting customer confidence and regulatory compliance. Technical files include complete test reports, calibration certificates, and quality assurance records.
Ongoing quality improvements incorporate feedback from distributors, healthcare providers, and end-users worldwide. This commitment to continuous improvement ensures MAIKONG quantum devices maintain competitive advantages in performance, reliability, and customer satisfaction.
Comprehensive labeling and documentation communicate essential information enabling safe and effective quantum device use. Regulatory frameworks specify mandatory content, format, and presentation requirements ensuring users receive necessary instructions and safety information.
Quantum device labels must include specific information elements identifying the manufacturer, device model, serial numbers, manufacturing dates, and applicable safety warnings. Standardized symbols communicate important information without language barriers, supporting international device distribution.
ISO 15223-1 establishes symbols used in medical device labeling. These symbols indicate electrical safety classifications, protection against water ingress, temperature limits, and handling precautions. Proper symbol usage ensures users worldwide understand critical device characteristics regardless of language differences.
Nigerian market requirements specify English language documentation. MAIKONG provides comprehensive English language materials including device labels, user manuals, quick start guides, and technical specifications. This documentation supports Nigerian healthcare provider training and ongoing device operation.

Comprehensive user manuals provide detailed instructions supporting safe and effective quantum device operation. Manual content includes device description, intended use statement, contraindications, operating instructions, maintenance procedures, troubleshooting guidance, and technical specifications.
Instructions must address reasonably foreseeable misuse scenarios warning users about improper operation potentially compromising safety or performance. Safety precautions highlight electrical safety requirements, patient positioning considerations, and environmental condition limitations affecting device operation.
MAIKONG user manuals incorporate visual aids, step-by-step procedures, and troubleshooting flowcharts supporting user comprehension. Downloadable manual formats enable convenient access supporting healthcare provider training and reference needs.
Technical documentation supports regulatory submissions, customer evaluations, and distributor technical knowledge requirements. Documentation packages include design specifications, test reports, clinical evaluation summaries, risk management files, and quality system certifications.
Quantum device specifications detail electrical characteristics, environmental operating ranges, software requirements, measurement parameters, analysis algorithms, and reporting capabilities. Complete specifications enable informed purchasing decisions and proper device integration into existing healthcare infrastructure.
Distributors receive comprehensive technical documentation packages supporting customer inquiries, regulatory submissions, and tender responses. MAIKONG maintains updated documentation reflecting ongoing product improvements and regulatory requirement changes, ensuring distributors always access current information.
Distributors assume significant compliance responsibilities when importing and selling medical quantum devices in Nigerian markets. Understanding these obligations protects distributors from regulatory penalties while ensuring end-users receive safe, effective, properly supported products.
Nigerian medical device importers must obtain appropriate licenses from NAFDAC before commencing distribution activities. Application processes require comprehensive documentation including business registration certificates, facility inspection reports, quality management system evidence, and technical staff qualification records.
Product registration represents a separate requirement from importer licensing. Each quantum device model requires individual registration involving technical documentation review, sample testing when applicable, and registration fee payment. Registration certificates authorize specific device models for Nigerian market distribution.
MAIKONG supports Nigerian distributors throughout registration processes. The company provides complete technical dossiers, certification documents, and regulatory correspondence supporting efficient NAFDAC submissions. This comprehensive support accelerates market entry while minimizing distributor administrative burdens.
Proper quantum device storage maintains product quality and safety throughout distribution chains. Storage facilities must provide appropriate environmental conditions including temperature control, humidity management, and protection from physical damage. Environmental monitoring records demonstrate ongoing compliance with storage requirements.
Handling procedures prevent damage during transportation and delivery activities. Packaging designs protect quantum devices from shock, vibration, and environmental exposure during shipping. Distributors must inspect devices upon receipt, verifying packaging integrity and documenting any apparent damage before accepting shipments.
Inventory management systems implement first-in-first-out rotation preventing prolonged storage potentially affecting device performance. Regular inventory audits verify physical stock matches records while identifying devices approaching expiration dates when applicable. Proper inventory management reduces waste while ensuring customers receive devices within optimal condition.

Distributors must ensure healthcare providers receive adequate training supporting safe and effective quantum device operation. Training programs cover device setup, patient preparation, operation procedures, result interpretation, maintenance requirements, and troubleshooting common issues.
Technical support services address customer questions, operational challenges, and maintenance needs throughout device lifecycle. Support systems include telephone helplines, email support, remote software assistance, and on-site service when necessary. Responsive technical support builds customer satisfaction and loyalty while preventing improper device use.
MAIKONG provides comprehensive distributor training programs covering product knowledge, technical specifications, regulatory compliance, and sales strategies. Nigerian distributors receive ongoing support including marketing materials, customer training resources, and technical troubleshooting assistance. This support infrastructure positions distributors for long-term business success.
Discover comprehensive support MAIKONG provides Nigerian distributors including regulatory documentation, technical training, marketing resources, and ongoing business development assistance. Join our growing network of successful West African partners.
MAIKONG maintains comprehensive international certifications supporting global quantum device distribution including Nigerian and broader African markets. This certification portfolio demonstrates commitment to quality, safety, and regulatory compliance across multiple jurisdictions.
MAIKONG Quantum Health Analyzer systems hold CE marking certification demonstrating conformity with European medical device directives. Notified body assessments verify quality management systems and technical documentation meet stringent European requirements. This certification facilitates market access across European Economic Area nations and provides credibility in international markets.
ISO 13485 certification validates MAIKONG quality management systems meet international standards for medical device manufacturers. Independent auditors verify comprehensive quality processes covering design, manufacturing, distribution, and servicing activities. Regular surveillance audits ensure ongoing compliance with evolving standard requirements.
Electrical safety certifications confirm MAIKONG quantum devices meet IEC 60601 requirements for medical electrical equipment. Testing reports document compliance with leakage current limits, insulation resistance specifications, electromagnetic compatibility requirements, and environmental condition performance. These certifications protect end-users while supporting distributor regulatory obligations.
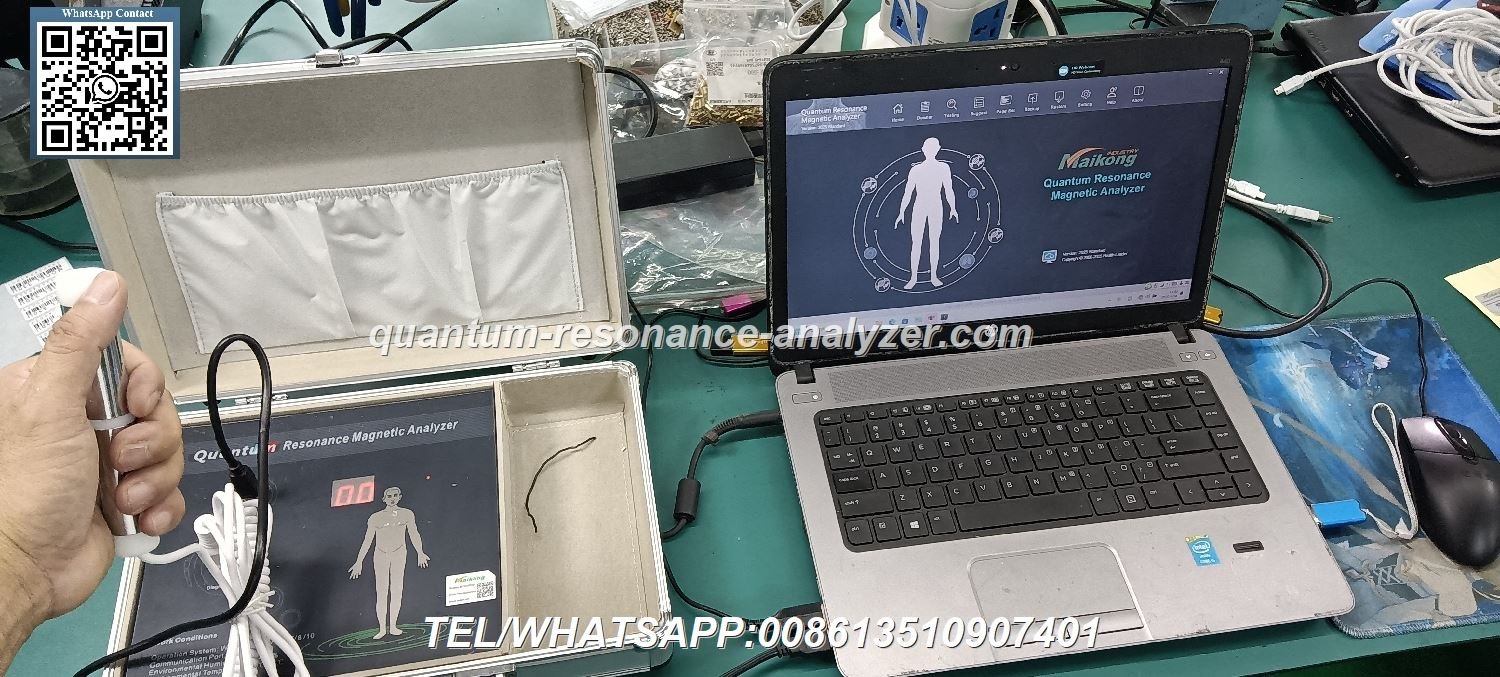
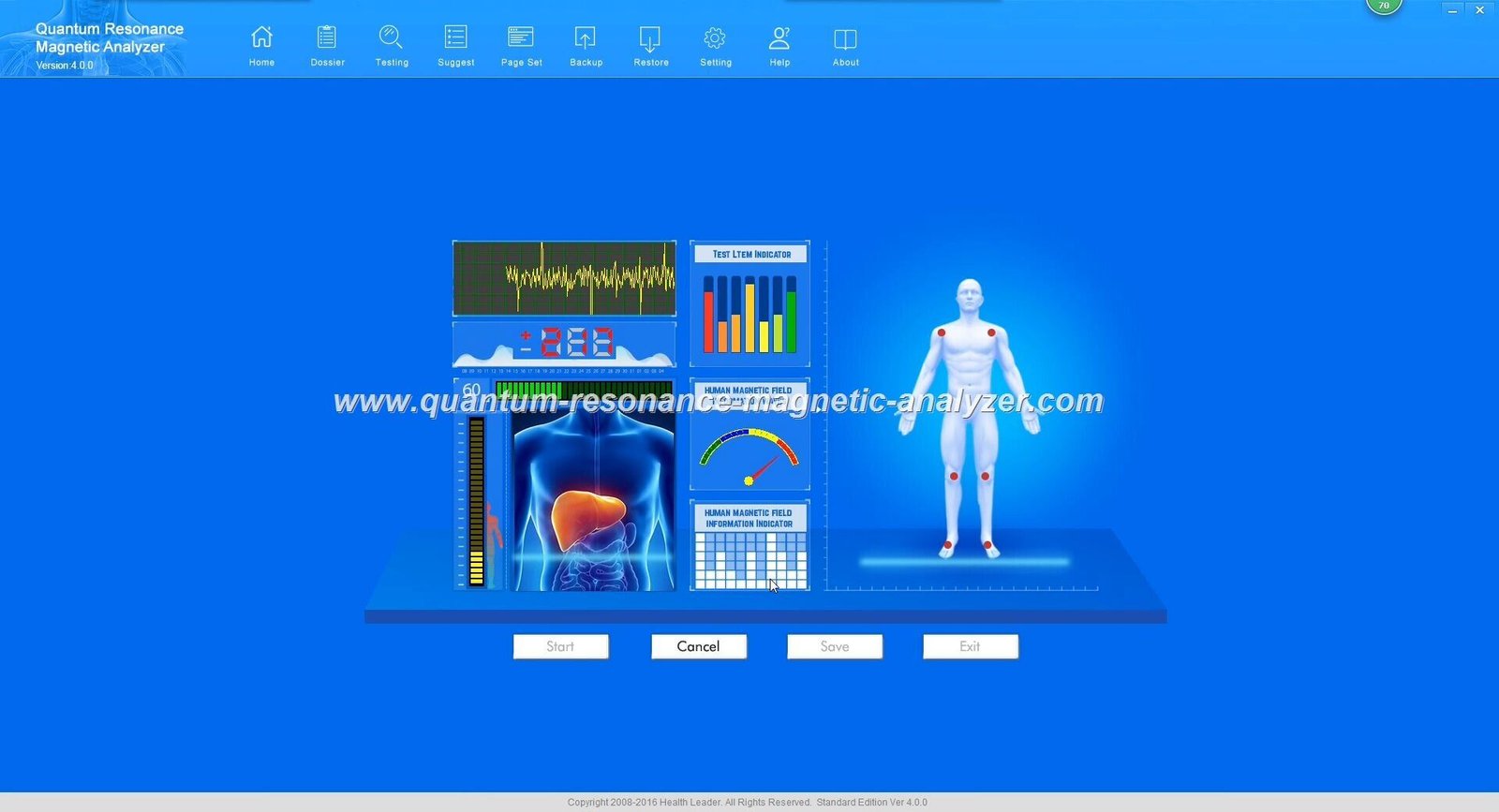
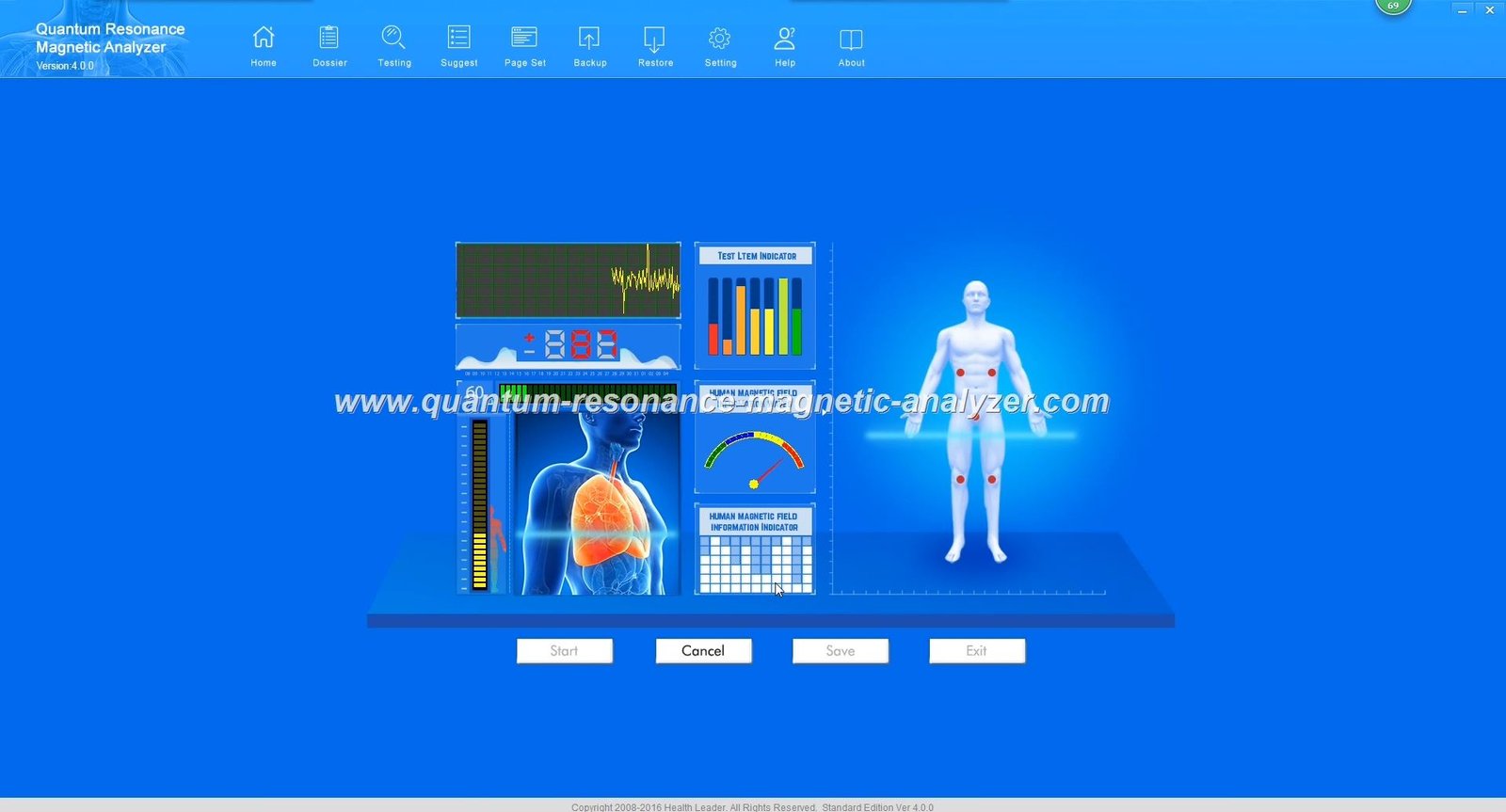
MAIKONG Quantum Analyzer represents advanced health analysis technology incorporating proven quantum resonance principles. The system analyzes electromagnetic frequencies emitted by human cells, processing this information through sophisticated quantum algorithms generating comprehensive health reports across fifty-four parameters.
Analysis procedures require approximately ninety seconds per patient. Users simply hold the hand sensor while the quantum device collects electromagnetic signals. Software processes this data displaying results through intuitive graphical interfaces. Report formats include both web-based displays and downloadable PDF documents supporting record-keeping and patient communication.
The quantum device operates across multiple language versions supporting international distribution. Available languages include English, Spanish, French, German, Polish, Portuguese, Malaysian, Indonesian, Bulgarian, Hungarian, Russian, Serbian, Slovak, Czech, Romanian, Vietnamese, Thai, Chinese, and Dutch. This multilingual capability enables deployment across diverse markets without requiring separate software versions.




MAIKONG software operates on Windows-based computer systems including Windows XP, Windows 7, Windows Vista, Windows 8, Windows 10, and Windows 11. This broad compatibility supports deployment in healthcare facilities with diverse existing infrastructure. The software does not support Android, Apple macOS, Linux, or Unix operating systems.
Minimum system requirements include computers with 2GB RAM, 500MB available storage space, and standard USB ports for device connectivity. These modest specifications enable operation on typical office computers without requiring specialized hardware investments. Installation procedures follow straightforward processes completable within minutes.
Analysis reports present comprehensive health information across cardiovascular function, gastrointestinal function, liver function, kidney function, lung function, brain nerve function, bone conditions, mineral levels, vitamin levels, amino acid levels, immune system status, and numerous additional parameters. Detailed report formats support healthcare provider interpretation and patient education.
Nigeria’s healthcare market presents substantial growth opportunities for certified medical quantum devices. Demographic trends, increasing health awareness, expanding middle class populations, and government healthcare initiatives create favorable conditions for innovative health analysis technology adoption.
Nigeria maintains Africa’s largest population exceeding 220 million people. Rapidly growing middle class segments increasingly seek preventive healthcare services and advanced diagnostic capabilities. Traditional healthcare infrastructure faces capacity challenges, creating opportunities for innovative solutions including non-invasive health analysis quantum devices.
Private healthcare facilities expand across major Nigerian cities including Lagos, Abuja, Port Harcourt, Kano, and Ibadan. These facilities invest in modern medical equipment enhancing service offerings and competitive positioning. Quantum health analyzers provide attractive value propositions combining advanced technology, non-invasive procedures, and comprehensive analysis capabilities.
Wellness centers, fitness facilities, pharmacies, and alternative medicine practitioners represent additional market segments. These businesses serve health-conscious consumers seeking preventive health assessments and lifestyle guidance. Quantum devices complement existing service offerings while generating new revenue streams through health analysis services.
MAIKONG distributors receive comprehensive support establishing profitable quantum device businesses in Nigerian markets. Partnership benefits include exclusive territorial rights, competitive wholesale pricing, complete technical documentation, ongoing training programs, marketing resource access, and responsive technical support services.
Initial distributor investments remain accessible for Nigerian entrepreneurs. Product orders start at modest quantities enabling market testing without excessive capital requirements. As distribution businesses grow, volume pricing structures provide increased profit margins supporting business expansion and market development activities.
Marketing support includes product brochures, demonstration videos, technical specification sheets, and digital marketing content. MAIKONG provides customizable materials enabling distributors to develop professional marketing campaigns targeting their specific customer segments. Social media content, email templates, and presentation materials accelerate market development efforts.
Primary customer segments for quantum device distribution include private hospitals, specialized clinics, wellness centers, diagnostic laboratories, corporate health programs, fitness centers, pharmacies with health services, alternative medicine practitioners, and government health initiatives. Each segment presents unique opportunities and requirements.
Private hospitals seek advanced diagnostic equipment enhancing service capabilities and patient satisfaction. Quantum devices provide comprehensive health screening services complementing traditional diagnostic methods. Non-invasive procedures appeal to patients while rapid analysis supports efficient clinic operations.
Corporate wellness programs increasingly incorporate preventive health assessments for employees. Quantum health analyzers deliver convenient, comprehensive screening supporting workplace wellness initiatives. Distributors can develop specialized packages targeting corporate clients seeking employee health services.
Successful quantum device deployment requires proper installation, comprehensive operator training, and ongoing technical support. MAIKONG provides complete support infrastructure ensuring Nigerian distributors and their customers achieve optimal device performance and user satisfaction.
Quantum device installation involves straightforward procedures completable within thirty minutes. The process includes unpacking equipment, connecting the hand sensor to the main processing unit, installing software on a Windows-based computer, and completing initial configuration settings.
Encryption lock activation protects software from unauthorized use while enabling legitimate users to operate quantum devices across multiple computers when needed. Activation procedures follow simple steps documented in user manuals and supported through technical assistance channels.
Environmental requirements remain minimal. Quantum devices operate effectively in standard office or clinical environments without special temperature control, humidity management, or electromagnetic shielding. Standard electrical outlets provide adequate power supply. USB connectivity enables operation with desktop or laptop computers meeting minimum system requirements.
Comprehensive operator training ensures healthcare providers use quantum devices effectively and interpret results appropriately. Training programs cover device operation, patient preparation, data collection procedures, report interpretation, maintenance requirements, and troubleshooting common issues.
Initial training sessions require approximately four hours covering all essential operational aspects. Hands-on practice sessions enable trainees to perform patient analyses under supervision, building confidence and competence. Training materials include written manuals, video demonstrations, and quick reference guides supporting ongoing learning.
Advanced training addresses result interpretation, clinical applications, and integration with other diagnostic methods. Healthcare professionals learn to correlate quantum device findings with patient symptoms, medical histories, and conventional diagnostic results. This comprehensive understanding maximizes clinical value delivered through quantum health analysis.
Ongoing technical support addresses operational questions, software issues, and hardware concerns throughout quantum device lifecycle. Support channels include telephone assistance, WhatsApp messaging, email correspondence, and remote computer access when needed. Nigerian distributors receive priority support ensuring rapid response to customer needs.
Preventive maintenance requirements remain minimal. Regular cleaning of the hand sensor maintains hygiene standards. Periodic software updates deliver performance improvements, additional features, and security enhancements. MAIKONG provides software updates free of charge to registered users maintaining current support agreements.
Hardware reliability ensures minimal downtime and repair requirements. Quality manufacturing processes and comprehensive testing produce durable quantum devices withstanding typical healthcare environment conditions. When repairs become necessary, MAIKONG provides efficient service restoring devices to full operational status.
MAIKONG provides Nigerian distributors and their customers with comprehensive training materials, video tutorials, technical documentation, and responsive support services. Ensure successful quantum device deployment and ongoing operational excellence.
Successful medical equipment distribution requires comprehensive business planning addressing market analysis, financial projections, marketing strategies, and operational infrastructure. Nigerian entrepreneurs entering quantum device distribution benefit from systematic planning approaches maximizing success probability while managing investment risks.
Effective market entry begins with target customer identification and segmentation. Nigerian healthcare markets include diverse customer types with varying needs, purchasing processes, and decision criteria. Focusing initial efforts on specific segments enables efficient resource deployment and accelerated market penetration.
Geographic focus strategies prove effective in large markets like Nigeria. Concentrating initial distribution efforts within specific cities or regions enables deeper market penetration and stronger customer relationships. Success in focused markets provides foundation for geographic expansion leveraging proven business models and customer testimonials.
Demonstration programs accelerate customer adoption by enabling healthcare providers to evaluate quantum devices before purchase commitments. Trial periods, satisfaction guarantees, and flexible payment terms reduce customer perceived risks. These strategies prove particularly effective when introducing innovative technologies to conservative healthcare markets.
Initial distributor investments include product inventory, marketing materials, demonstration equipment, business registration fees, and working capital supporting initial operations. MAIKONG’s flexible order quantities enable Nigerian entrepreneurs to begin distribution businesses with manageable initial investments ranging from modest to substantial based on individual business strategies.
Profit margins on quantum device sales support sustainable distribution businesses while maintaining competitive customer pricing. Volume pricing structures reward business growth with increased margins supporting expanded marketing investments and operational infrastructure. Recurring revenue opportunities through consumables, software updates, and service contracts enhance long-term business profitability.
Financial projections should model various growth scenarios from conservative to optimistic. Sensitivity analyses examine how changing assumptions about sales volumes, pricing levels, and cost structures affect financial outcomes. Conservative planning ensures business sustainability even if growth rates lag optimistic projections.

Integrated marketing approaches combining digital and traditional channels prove most effective for medical equipment distribution. Digital strategies include professional websites, search engine optimization, social media marketing, and email campaigns. Traditional approaches encompass trade show participation, professional journal advertising, and direct sales presentations.
Content marketing establishes distributor expertise and credibility. Educational articles, case studies, video demonstrations, and webinar presentations attract healthcare providers seeking quantum device information. This approach generates qualified leads more receptive to sales conversations than cold prospecting methods.
Professional networking within healthcare communities builds relationships supporting long-term business development. Participation in medical associations, healthcare conferences, and professional education events creates visibility and credibility. Relationships developed through networking often yield valuable referrals and partnership opportunities.
Distributors receive comprehensive marketing resources including product brochures, technical specification sheets, demonstration videos, and digital content.
Customizable materials enable distributors to develop professional marketing campaigns while maintaining brand consistency.
Social media templates, email campaign content, and presentation materials accelerate marketing program development.
Ongoing content updates ensure distributors access current information reflecting product improvements and expanded capabilities.
Regulatory compliance represents ongoing obligations rather than one-time achievements. Distributors must maintain current registrations, implement post-market surveillance systems, respond to regulatory inquiries, and adapt to evolving requirements ensuring continued market authorization for quantum devices.
Post-market surveillance systems monitor quantum device performance throughout commercial distribution. These systems collect information from multiple sources including customer complaints, service records, adverse event reports, and periodic safety updates. Systematic analysis identifies trends potentially indicating safety or performance concerns requiring investigation.
Adverse event reporting obligations require distributors to document and report serious incidents to regulatory authorities within specified timeframes. Serious incidents include device malfunctions potentially causing patient harm, deaths or serious injuries potentially related to device use, and significant performance failures affecting diagnostic accuracy.
MAIKONG maintains comprehensive global vigilance systems supporting distributor compliance obligations. The company investigates reported incidents, implements corrective actions when needed, and communicates relevant information to distributors and regulatory authorities. This systematic approach protects patients while demonstrating commitment to product safety and regulatory compliance.
Medical device regulations evolve continuously as authorities incorporate new scientific evidence, address emerging technologies, and harmonize international requirements. Distributors must monitor regulatory developments affecting quantum device compliance obligations and implement necessary changes maintaining market authorization.
NAFDAC periodically updates registration requirements, safety standards, and compliance expectations. Registration renewals provide opportunities for regulatory authorities to verify ongoing compliance with current requirements. Distributors should maintain regular communication with NAFDAC understanding upcoming changes and planning appropriate responses.
MAIKONG monitors global regulatory developments affecting quantum device requirements. The company proactively updates products, documentation, and compliance programs addressing evolving standards. Distributors receive timely notifications about regulatory changes and guidance implementing necessary updates ensuring continued compliance.

Distributors implementing quality management systems benefit from improved operational efficiency, reduced errors, and enhanced customer satisfaction. Quality systems establish documented procedures for key business processes including order processing, inventory management, customer training, technical support, and complaint handling.
Regular internal audits verify quality system effectiveness and identify improvement opportunities. Audit findings drive corrective actions addressing identified deficiencies. Management reviews examine audit results, customer feedback, and performance metrics determining strategic quality objectives and resource allocation priorities.
Quality system maturity develops progressively. Initial implementations focus on essential processes supporting regulatory compliance. As distributor organizations mature, quality systems expand addressing additional business processes and incorporating advanced quality management tools. This progressive approach balances compliance requirements with business development priorities.
Nigerian quantum device distributors should establish systematic processes addressing these ongoing compliance obligations:
MAIKONG quantum devices deliver distinct competitive advantages supporting distributor market success. These advantages span product performance, certification completeness, technical support quality, business partnership terms, and ongoing innovation commitment.
MAIKONG Quantum Analyzer systems demonstrate consistent performance across diverse operating environments and patient populations. Rigorous manufacturing quality controls and comprehensive testing protocols ensure every device meets specifications before shipment. Field reliability data confirms low failure rates and minimal maintenance requirements supporting customer satisfaction.
Analysis accuracy represents critical performance characteristics for diagnostic quantum devices. MAIKONG validation studies document high correlation between device measurements and reference methods across multiple health parameters. Reproducibility testing confirms consistent results when measuring the same subjects repeatedly under controlled conditions.
User interface design prioritizes intuitive operation minimizing training requirements and operational errors. Clear visual displays, logical menu structures, and helpful prompts guide operators through analysis procedures. Report formats present comprehensive information through professional layouts supporting healthcare provider interpretation and patient communication.
Complete certification portfolios differentiate MAIKONG from competitors offering products with limited or unclear regulatory status. CE marking, ISO 13485 certification, electrical safety compliance, and software validation documentation provide objective evidence of product quality and safety supporting customer confidence and regulatory compliance.
Technical documentation completeness enables efficient regulatory submissions, tender responses, and customer evaluations. MAIKONG provides comprehensive technical files including design specifications, test reports, clinical evaluations, risk management documentation, and quality system certificates. This documentation supports distributor business development activities and regulatory obligations.
Nigerian-specific documentation addresses local market requirements including English language materials, NAFDAC registration support, and regional technical support information. This localization demonstrates MAIKONG commitment to Nigerian market success while simplifying distributor compliance processes.
MAIKONG distributor partnerships extend beyond simple product supply relationships. The company provides comprehensive business support including market development guidance, marketing resources, technical training, ongoing support services, and responsive communication. This partnership approach positions distributors for long-term business success.
Competitive pricing structures enable distributors to offer attractive customer pricing while maintaining healthy profit margins. Volume pricing rewards business growth with improved margins supporting expanded marketing investments. Flexible payment terms accommodate distributor cash flow requirements during business development phases.
Exclusive territorial arrangements protect distributor investments by preventing internal competition. Territory definitions align with distributor market development capabilities and business plans. Clear territorial boundaries enable focused market development strategies maximizing return on distributor time and resource investments.
MAIKONG welcomes qualified Nigerian entrepreneurs, medical equipment distributors, and healthcare business professionals to join our growing West African distribution network. The distributor application process involves several steps ensuring mutual understanding of expectations, capabilities, and partnership objectives.
Successful distributor candidates demonstrate several key qualifications. Business registration and legal compliance provide foundation for professional operations. Healthcare industry experience or strong business development backgrounds indicate capability to understand customer needs and communicate product benefits effectively.
Financial capacity to invest in initial inventory, marketing programs, and business infrastructure represents important considerations. While MAIKONG accommodates various business scales through flexible order quantities, distributors must demonstrate adequate resources supporting sustained business development efforts.
Market knowledge and customer relationships within Nigerian healthcare communities provide significant advantages. Established networks accelerate market penetration and customer acquisition. However, motivated entrepreneurs without extensive healthcare industry experience can succeed through dedication and systematic market development approaches.
Initial inquiries begin through direct contact via WhatsApp, email, or telephone. MAIKONG representatives discuss distributor interest, business backgrounds, target markets, and preliminary questions. This initial conversation provides mutual understanding determining whether detailed discussions warrant investment of both parties’ time.
Qualified candidates receive comprehensive distributor information packages including product specifications, certification documentation, pricing structures, territory definitions, partnership terms, and business development resources. Candidates review these materials considering how quantum device distribution aligns with their business objectives and capabilities.
Subsequent discussions address specific questions, clarify partnership terms, and explore market development strategies. MAIKONG representatives provide guidance addressing regulatory requirements, competitive positioning, and business planning considerations. These consultative discussions help candidates develop realistic business plans and success strategies.

Formal partnership agreements document distributor rights, responsibilities, territory definitions, pricing structures, and support commitments. Clear written agreements prevent misunderstandings while providing frameworks for successful long-term partnerships. Legal review of partnership agreements ensures both parties understand and accept terms.
Initial orders include training and launch support ensuring successful market entry. MAIKONG provides product training, sales technique guidance, marketing material customization, and technical support orientation. This comprehensive onboarding positions new distributors for effective customer engagement from business launch.
Ongoing partnership management includes regular communication, performance reviews, and continuous improvement initiatives. MAIKONG maintains accessible communication channels supporting distributor questions, market development discussions, and problem resolution. This sustained engagement builds strong partnerships delivering mutual success.
Join MAIKONG’s successful West African distribution network. Access certified products, comprehensive support, and profitable business opportunities serving Nigeria’s growing healthcare market. Contact us today to begin your distributor application process.
Complete the form below to begin your MAIKONG quantum device distributor application. Our partnership team will review your information and contact you within forty-eight hours to discuss distribution opportunities in your target market.
Nigerian entrepreneurs and healthcare professionals frequently ask similar questions about quantum device distribution, regulatory requirements, and partnership opportunities. This section addresses common inquiries providing helpful information for prospective distributors.
MAIKONG Quantum Analyzer holds comprehensive international certifications including CE marking demonstrating European conformity, ISO 13485 quality management system certification, IEC 60601 electrical safety compliance, and IEC 62304 software lifecycle validation. These certifications support NAFDAC registration processes required for Nigerian market distribution. Complete certification documentation accompanies distributor packages enabling efficient regulatory submissions.
NAFDAC registration timelines vary based on application completeness and current agency workload. Well-prepared submissions with complete technical documentation typically receive approval within three to six months. MAIKONG provides comprehensive documentation packages supporting efficient registration processes. Our experience supporting Nigerian distributor registrations enables us to guide partners through requirements minimizing delays and maximizing approval probability.
Initial investment requirements depend on distributor business scale and market strategy. MAIKONG accommodates various business models through flexible minimum order quantities. Initial investments typically include product inventory for demonstrations and initial sales, marketing materials, business registration fees, and working capital supporting operations during market development phases. Specific investment details are discussed during distributor qualification conversations allowing customization to individual circumstances.
Yes, MAIKONG offers exclusive territorial distribution agreements to qualified partners. Territory definitions align with distributor market development capabilities and business plans. Exclusive arrangements protect distributor investments by preventing internal competition enabling focused market development strategies. Territory boundaries and exclusivity terms are documented in formal partnership agreements providing clear understanding of rights and responsibilities.
MAIKONG provides comprehensive distributor support including initial product training covering technical specifications and operation procedures, sales technique guidance for effective customer engagement, marketing resource access including brochures and digital content, technical support orientation covering customer service procedures, and ongoing partnership management through regular communication. This support infrastructure positions distributors for successful market entry and sustained business growth. Contact our partnership team for detailed information about specific training programs and support services.
Yes, MAIKONG Quantum Analyzer incorporates universal power supplies supporting voltage ranges from 110 to 240 volts and frequencies of 50 to 60 hertz. Nigerian electrical systems typically provide 230 volts at 50 hertz frequency falling within device operating specifications. Built-in protection circuits guard against voltage fluctuations and power surges common in developing market electrical infrastructure. No external voltage converters or special electrical installations are required for Nigerian deployment.
MAIKONG Quantum Analyzer software supports over twenty international languages including English, Spanish, French, German, Polish, Portuguese, Malaysian, Indonesian, Bulgarian, Hungarian, Russian, Serbian, Slovak, Czech, Romanian, Vietnamese, Thai, Chinese, and Dutch. English language versions serve Nigerian markets where English represents the primary business and professional communication language. Multiple language support enables distributors serving diverse international customer bases or expanding into neighboring West African markets.
Quantum resonance analysis provides complementary health assessment capabilities rather than replacing conventional diagnostic methods. The technology analyzes electromagnetic frequencies emitted by human cells generating comprehensive health status reports across fifty-four parameters within ninety seconds. This rapid, non-invasive screening identifies potential health concerns warranting further investigation through conventional diagnostic procedures. Healthcare providers use quantum analysis for preventive health screening, wellness assessments, and monitoring health status changes over time. The approach complements rather than replaces established diagnostic methods providing additional information supporting comprehensive patient care.
Specific profit margin information is provided during distributor qualification discussions as margins depend on factors including order volumes, territory characteristics, and competitive positioning. MAIKONG structures wholesale pricing enabling distributors to maintain healthy profit margins while offering competitive customer pricing. Volume pricing rewards business growth with improved margins supporting expanded marketing investments. Recurring revenue opportunities through consumables, software updates, and service contracts enhance long-term business profitability beyond initial device sales.
Timeline for beginning active sales depends on several factors including NAFDAC registration completion, initial inventory receipt, and market preparation activities. Distributors can begin market development activities including customer education, demonstration programs, and relationship building while registration processes proceed. Upon registration approval and inventory receipt, trained distributors can commence immediate sales activities. MAIKONG supports accelerated market entry through comprehensive onboarding programs and responsive communication throughout launch phases. Most distributors achieve first sales within three to six months from initial partnership establishment.
International safety requirements for quantum devices establish comprehensive frameworks protecting patients, healthcare providers, and end-users worldwide. Understanding these requirements enables Nigerian distributors to select certified products, navigate regulatory compliance, and build customer trust supporting long-term business success.
MAIKONG Quantum Health Analyzer represents proven technology meeting rigorous international safety standards including CE marking, ISO 13485 certification, electrical safety compliance, and software validation. These certifications combined with comprehensive distributor support position Nigerian partners for successful market entry and sustained business growth.
Nigeria’s expanding healthcare market presents substantial opportunities for innovative diagnostic technologies. Growing populations, increasing health awareness, expanding private healthcare infrastructure, and supportive government policies create favorable conditions for quantum device adoption across multiple customer segments.
Successful distribution requires more than quality products and certifications. Distributors must implement comprehensive business strategies addressing market analysis, financial planning, marketing development, regulatory compliance, technical support, and customer relationship management. MAIKONG partnership support provides resources, guidance, and ongoing assistance enabling distributor success.
The time for action arrives now. Nigerian healthcare markets continue evolving creating opportunities for early entrants establishing strong market positions. MAIKONG welcomes qualified partners joining our West African distribution network accessing certified products, proven business models, and comprehensive support infrastructure.

Join successful Nigerian entrepreneurs building profitable healthcare technology businesses with MAIKONG partnership support. Access certified quantum devices, comprehensive training, marketing resources, and ongoing technical assistance. Contact us now to begin your distributor qualification process and receive detailed partnership information.
Take the next step toward building your successful medical equipment distribution business. MAIKONG provides everything needed for market entry success:
Contact Information:
WhatsApp/Phone: +86-135-1090-7401
Email: Lucy@quantumanalyzer.ng
Website: https://quantumanalyzer.ng
