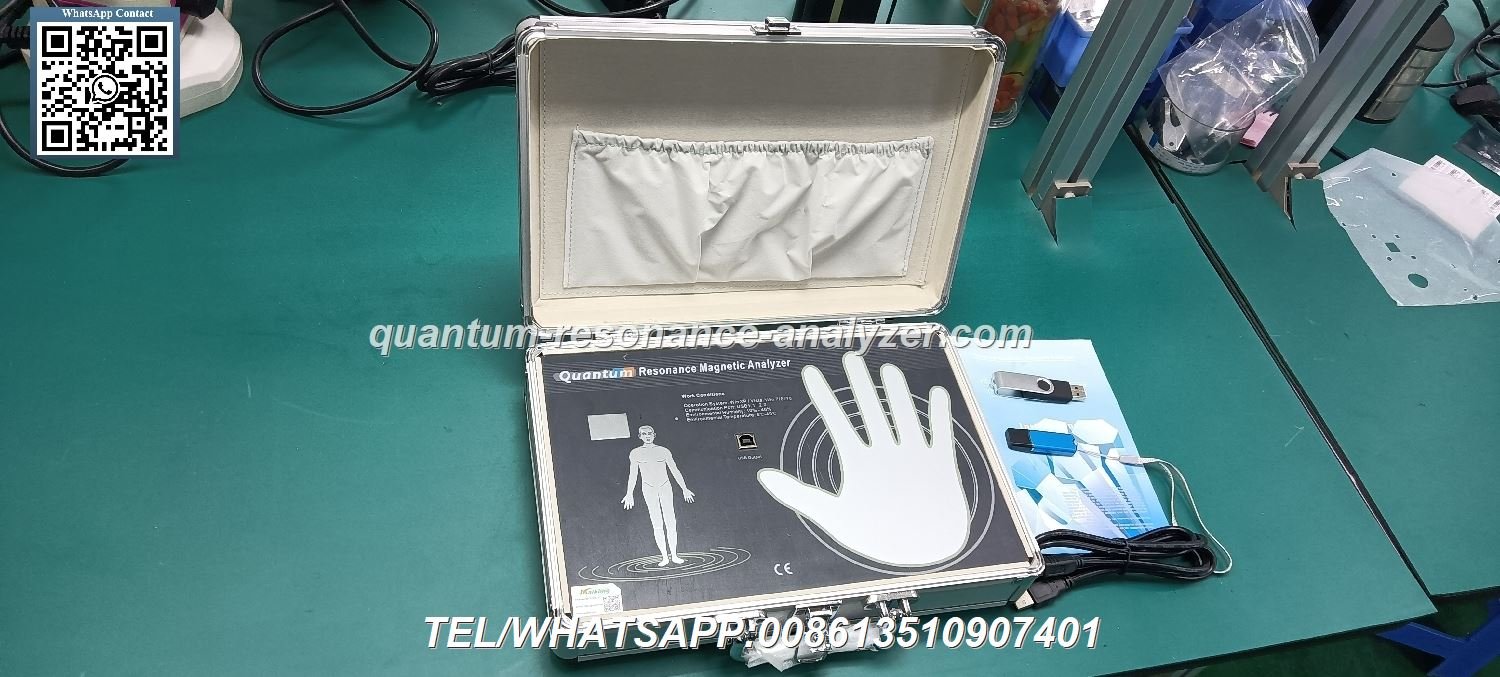
MAIKONG Certifications to Look for When Purchasing a Quantum Health Analyzer
Verify MAIKONG Certifications
Explore our complete certification documentation and quality assurance processes that set MAIKONG apart in the Nigerian market.
Why Certifications Matter When Buying a Quantum Health Analyzer

Medical equipment certifications serve as your first line of defense. They guarantee that a device meets strict safety protocols. International certification bodies conduct rigorous testing before approval. Without these seals, you gamble with patient safety.
The quantum resonance analyzer performs non-invasive body health assessments. It generates detailed reports about organ function and wellness markers. Inaccurate readings from uncertified machines can mislead treatment decisions. This creates liability issues for health practitioners.
Nigerian healthcare regulations increasingly emphasize quality standards. Government agencies scrutinize imported medical equipment. Certified devices clear customs smoothly and meet local compliance requirements. They also command higher trust from educated patients.
Benefits of Certified Equipment
- Legal compliance with Nigerian healthcare regulations
- Enhanced patient trust and confidence
- Reliable accuracy in health analysis reports
- Manufacturer accountability and support
- Higher resale value for equipment
- Reduced malpractice liability
Risks of Uncertified Devices
- Potential customs seizure during import
- Inaccurate diagnostic results
- No manufacturer warranty coverage
- Legal exposure in patient disputes
- Difficulty obtaining insurance
- Poor long-term reliability
Certifications also reflect manufacturing excellence. Companies that invest in compliance demonstrate commitment to quality. They maintain proper quality control systems throughout production. This translates to longer device lifespan and consistent performance.
MAIKONG prioritizes certification across our entire product line. Our quantum health analyzer carries both CE and ISO certifications. We maintain transparent documentation for all quality claims. Nigerian distributors receive complete certification packages with each order.
Connect with MAIKONG Nigeria Team
Speak directly with our certification specialists about quality standards and distributor opportunities in your region.
CE Certification for Quantum Health Analyzer Devices

The CE mark represents European Conformity standards. It indicates that a product meets EU safety requirements. For medical devices like the quantum resonance magnetic analyzer, CE certification is essential. This certification covers electrical safety, electromagnetic compatibility, and clinical performance.
Obtaining CE approval requires extensive documentation. Manufacturers must prove their device performs as claimed. They submit technical files including risk analysis and clinical data. Third-party notified bodies audit the manufacturing process. Only compliant products receive the coveted CE mark.
What CE Certification Covers for Health Analyzers
CE certification for a quantum analyzer machine addresses multiple safety aspects. Electrical components must meet strict insulation standards. The device cannot emit harmful electromagnetic radiation. Software algorithms require validation against medical standards.
The certification also mandates proper labeling and user instructions. Manufacturers must provide clear usage guidelines. They document potential risks and contraindications. This ensures health practitioners use the equipment safely.
- Electrical safety compliance with EN 60601 standards
- Electromagnetic compatibility testing
- Biocompatibility of patient contact materials
- Software validation and clinical performance verification
- Risk management documentation
- Post-market surveillance requirements
How to Verify CE Certification Authenticity

Unfortunately, fake CE marks exist in the marketplace. Unscrupulous sellers print the logo without proper authorization. Smart buyers verify certification through official channels. Request the full Declaration of Conformity description from your supplier.
Authentic CE certificates include specific details. Look for the notified body identification number. Check that the certificate covers your exact device model. Cross-reference the manufacturer details against official registries. MAIKONG provides complete CE documentation with transparent notified body information.
Verification Tip: Genuine CE certificates include a four-digit notified body number after the CE mark. Devices in Class I or higher classifications require this number. Always request this documentation before purchase.
Contact the manufacturer directly to confirm certification status. Legitimate companies readily share their compliance documents. They provide technical files and test reports upon request. Hesitation or vague responses signal potential problems.
ISO Certification Standards for Quantum Health Analyzer Manufacturing

ISO certifications focus on quality management systems. They ensure consistent manufacturing processes. For medical equipment, ISO 13485 represents the gold standard. This certification governs the entire production lifecycle of health devices.
Companies with ISO 13485 certification maintain rigorous quality controls. They document every production step. Regular audits verify continued compliance. This systematic approach minimizes defects and ensures reliable product performance.
ISO 13485 and Medical Device Quality
ISO 13485 differs from general quality standards. It specifically addresses medical device manufacturing requirements. The standard covers design controls and supplier management. It mandates proper documentation and traceability systems. Manufacturers must implement corrective action procedures.
This certification benefits end users significantly. Devices from ISO 13485 facilities demonstrate consistent quality. Manufacturing processes undergo continuous improvement. The quantum resonance analyzer you receive today matches tomorrow’s quality level.
Key ISO 13485 Requirements
- Design and development validation
- Supplier quality management
- Production process controls
- Equipment maintenance and calibration
- Employee training documentation
- Complaint handling procedures
- Internal audit systems
Additional ISO Certifications to Consider
Beyond ISO 13485, other certifications indicate manufacturer commitment. ISO 9001 covers general quality management principles. While less specific than 13485, it demonstrates organizational quality culture. Some manufacturers also hold ISO 14001 for environmental management.
MAIKONG maintains both ISO 13485 and ISO 9001 certifications. Our manufacturing facility in Guangdong operates under these standards. We conduct annual surveillance audits to maintain compliance. Nigerian distributors benefit from this quality assurance infrastructure.
“ISO certification isn’t just a badge. It represents a commitment to consistent excellence that Nigerian health practitioners can rely on throughout their device ownership.”
FDA Registration and Other Important Certifications for Health Analyzers

While CE and ISO form the foundation, additional certifications enhance credibility. FDA registration applies to devices sold in the United States market. Though not mandatory for Nigerian sales, it signals superior quality standards. FDA-registered manufacturers meet exceptionally high requirements.
The FDA registration process involves facility inspection. Manufacturers must demonstrate Good Manufacturing Practices (GMP). They maintain detailed device listings and reporting systems. This level of scrutiny ensures top-tier manufacturing standards.
Understanding FDA Registration for Quantum Analyzers
FDA registration differs from FDA approval or clearance. Registration means the manufacturer and facility are officially listed. The specific device may not require pre-market approval. However, registration still indicates regulatory compliance and quality consciousness.
For Nigerian buyers, FDA registration provides additional confidence. It shows the manufacturer operates at international standards. Devices acceptable in the strict U.S. market typically exceed requirements elsewhere. This matters when building a reputation for quality care.
Country-Specific Certifications to Consider
Different markets require different certifications. Australia demands TGA registration. Canada requires Health Canada approval. China has CFDA requirements for domestic sales. Understanding these helps evaluate manufacturer global reach.
MAIKONG products hold certifications across multiple jurisdictions. Our quantum health analyzer meets standards in North America, Europe, and Asia. This global acceptance demonstrates our commitment to universal quality standards.
| Certification | Issuing Authority | Scope | Importance Level |
| CE Mark | European Union Notified Bodies | Safety, Performance, Quality | Essential |
| ISO 13485 | International Organization for Standardization | Quality Management System | Essential |
| FDA Registration | U.S. Food and Drug Administration | Facility and Device Listing | Highly Recommended |
| ISO 9001 | International Organization for Standardization | General Quality Management | Recommended |
| TGA Registration | Australian Therapeutic Goods Administration | Australian Market Compliance | Optional |
Request MAIKONG Certification Package
Download our complete certification documentation including CE certificates, ISO compliance records, and international registration details.
Software Certification and Data Security for Quantum Resonance Magnetic Analyzer
Hardware certifications only tell part of the story. The software powering your quantum resonance magnetic analyzer requires equal attention. Medical software must meet strict data security standards. Patient information demands robust protection.
Modern quantum analyzers generate detailed health reports. This data contains sensitive personal information. Proper encryption prevents unauthorized access. Software certification ensures data handling meets privacy regulations.
Software Validation and Medical Device Standards
Medical device software requires validation under IEC 62304 standards. This framework governs software lifecycle processes. Developers must document all code changes. Testing protocols verify functionality under all conditions. Risk management identifies potential software failures.
MAIKONG quantum resonance analyzer software undergoes rigorous validation. We maintain version control and change documentation. Our software supports Windows XP through Windows 11. This compatibility ensures long-term usability across various home and clinical environments.
- IEC 62304 software lifecycle compliance
- Encryption protocols for patient data storage
- Secure user authentication systems
- Regular software security updates
- Backup and recovery features
- Audit trail functionality for clinical environments
Encryption Lock Technology

MAIKONG implements encryption lock technology for device security. Each quantum health analyzer ships with a unique USB encryption key. This prevents unauthorized software use and protects your investment. The system ensures only legitimate licensed users access the device.
The encryption lock also ties software updates to authorized users. You receive guaranteed update access throughout your warranty period. This security feature prevents counterfeit software proliferation. Nigerian distributors appreciate this protection of their business interests.
Security Note: Always verify that your quantum analyzer includes an original encryption lock. This USB key authenticates genuine MAIKONG software and ensures you receive manufacturer support and updates.
Our comprehensive encryption lock guide provides setup instructions. Nigerian users can access detailed documentation in English. The system works seamlessly on all supported Windows operating systems. We offer technical support through WhatsApp for any configuration questions.
Warranty and After-Sales Support as Quality Indicators

Certifications validate initial quality. Warranty terms reveal long-term confidence. Manufacturers who stand behind their products offer comprehensive warranties. The warranty period and coverage terms indicate expected reliability.
MAIKONG provides a standard one-year warranty on all quantum health analyzer devices. This coverage includes manufacturing defects and component failures. We replace or repair defective units promptly. Our warranty reflects confidence in our quality control processes.
Understanding Warranty Coverage
Read warranty terms carefully before purchase. Some warranties cover parts only. Others include labor for repairs. Shipping costs may or may not be included. Understanding these details prevents surprises later.
Our warranty covers the complete device including hand sensors and cables. We provide software updates throughout the warranty period. Technical support remains available via online channels. Nigerian distributors receive priority support for their customers.
MAIKONG Warranty Includes
- 12-month comprehensive coverage
- Free software updates and patches
- Online technical support
- Replacement parts for defects
- Email and WhatsApp support channels
- English language documentation
Warranty Exclusions
- Physical damage from misuse
- Water or liquid damage
- Unauthorized modifications
- Normal wear and tear items
- Lost encryption keys
- Software installation on unsupported OS
The Importance of Technical Support

After-sales service quality matters as much as initial product quality. Technical questions arise during device setup. Users need guidance interpreting reports. Responsive support makes the difference between satisfaction and frustration.
MAIKONG maintains dedicated support channels for Nigerian customers. We offer online technical support through multiple platforms. Our team understands the unique challenges of the Nigerian market. Response times typically occur within 24 hours during business days.
We also provide comprehensive documentation resources. The quantum resonance magnetic analyzer manual covers all operational aspects. Nigerian distributors receive training materials for end customers. This support infrastructure enables successful long-term device use.
Download Free User Manual
Access the complete quantum resonance magnetic analyzer user manual in English. Learn proper operation, maintenance, and troubleshooting procedures.
Special Considerations for Nigerian Buyers of Health Analyzers

The Nigerian health market presents unique opportunities and challenges. Rapid economic growth drives demand for quality medical equipment. Yet import regulations and logistics require careful navigation. Understanding local requirements ensures smooth business operations.
Certified devices clear Nigerian customs more easily. Proper documentation prevents delays. Working with experienced suppliers like MAIKONG simplifies the import process. We provide all necessary certification paperwork for Nigerian shipping.
Import Requirements and Customs Clearance
Nigerian authorities require specific documentation for medical equipment imports. CE and ISO certificates form part of this paperwork. Commercial invoices must accurately describe the product. Customs officials may request additional technical specifications.
MAIKONG prepares complete documentation packages for Nigerian shipments. We include all certification copies and technical files. Our experience with Nigerian customs reduces clearance time. Distributors receive guidance on local registration requirements.
- CE certificate copies for customs review
- ISO certification documentation
- Detailed product specifications and technical files
- Commercial invoice with accurate HS codes
- Bill of lading and shipping documentation
- Packing list with detailed contents
Building a Successful Distribution Business

Many Nigerian entrepreneurs seek profitable health care opportunities. Becoming a MAIKONG distributor offers excellent potential. The quantum health analyzer serves multiple market segments. Clinics, wellness centers, and pharmacies all represent potential customers.
Successful distributors understand local market needs. They provide training for end users. Technical support builds customer loyalty. MAIKONG supports our Nigerian partners with marketing materials and training resources.
Medical Clinics

Private clinics seek advanced diagnostic equipment. The quantum analyzer provides comprehensive health screening capabilities. It enhances service offerings and attracts health-conscious patients.
Wellness Centers

Wellness practitioners focus on preventive health. Non-invasive body scanning aligns with holistic approaches. The detailed reports support personalized wellness plans and supplement recommendations.
Pharmacy Chains

Forward-thinking pharmacies add value-added services. Health screening attracts customers and increases foot traffic. The analyzer generates upsell opportunities for supplements and wellness products.
Financing and Payment Options
We understand that establishing a distribution business requires investment. MAIKONG offers flexible payment terms for qualified Nigerian distributors. We accept multiple payment methods including bank transfer and credit cards. Bulk order discounts incentivize larger initial investments.
Starting with a smaller order allows market testing. Many successful distributors began with 5-10 units. As demand grows, they scale their operations. We support this gradual expansion approach with consistent product availability.
Distributor Opportunity: MAIKONG seeks dedicated partners throughout Nigeria. We offer territorial protection, marketing support, and comprehensive training. Minimum order quantities start at just 5 units. Contact us to discuss becoming an authorized distributor in your region.
How to Verify Quantum Health Analyzer Authenticity and Certifications

Counterfeit medical devices plague the global market. Unscrupulous sellers copy popular products. They print fake certification marks. These imitations lack proper quality control and safety testing. Protecting yourself requires vigilance.
Legitimate manufacturers provide verification methods. Serial numbers enable authenticity checks. Certification documents should reference specific device models. Packaging quality often reveals counterfeit products. MAIKONG implements multiple anti-counterfeiting measures.
Steps to Verify Your Device
- Request complete certification documentation before purchase
- Verify CE certificate notified body number against official EU databases
- Check ISO certification validity through issuing registrar websites
- Confirm serial number with manufacturer directly
- Inspect packaging quality and holographic security features
- Verify software includes genuine encryption lock
- Contact manufacturer for authentication confirmation
MAIKONG registers all serial numbers in our system. Nigerian customers can contact us directly for verification. We confirm whether a serial number corresponds to a genuine MAIKONG unit. This service protects buyers from counterfeit devices.
Red Flags to Watch For
Certain warning signs indicate potential fraud. Prices significantly below market rates raise suspicion. Sellers who avoid certification questions likely sell counterfeits. Missing encryption locks or generic packaging signal problems. Always purchase from authorized distributors.
Warning Signs of Counterfeits: Be cautious of sellers offering “similar quality” at 30-50% lower prices, those unable to provide original CE certificates, devices lacking serial numbers, missing MAIKONG branding, or software without encryption locks. These indicate counterfeit products.
- Unusually low pricing compared to authorized distributors
- Inability to provide original certification documents
- Generic or poor-quality packaging materials
- Missing serial numbers or non-registered serial numbers
- Software without proper encryption lock
- Seller reluctance to allow manufacturer verification
- Vague or inconsistent warranty terms
MAIKONG takes intellectual property protection seriously. We pursue legal action against counterfeiters. Our authorized Nigerian distributors receive genuine products with full certification. Building relationships with verified suppliers protects your investment and reputation.
Verify Your Device Authenticity
Contact MAIKONG directly to authenticate any quantum health analyzer. We verify serial numbers and confirm genuine product status within 24 hours.
MAIKONG Quantum Health Analyzer Certification Credentials

MAIKONG has earned recognition as a leading quantum health analyzer manufacturer. Our 11-year manufacturing history demonstrates commitment to excellence. We maintain certifications across multiple international standards. Nigerian distributors benefit from our established quality reputation.
Our Guangdong facility operates under ISO 13485 quality management systems. Regular third-party audits verify continued compliance. We invest continuously in quality improvement initiatives. This dedication translates to reliable products for your business.
Our Certification Portfolio
MAIKONG holds comprehensive certifications for our quantum resonance magnetic analyzer product line. Each certification validates different quality aspects. Together, they provide complete assurance of product excellence. We share certification copies transparently with all distributors.
What Sets MAIKONG Apart

Many companies claim quality. Few demonstrate it through comprehensive certification. MAIKONG differentiates through transparent quality documentation. We maintain detailed records of all manufacturing processes. Our traceability systems track components from suppliers through device assembly.
We also invest heavily in research and development. Our engineering team continuously improves device features. Software updates enhance functionality over time. This commitment to innovation keeps MAIKONG products at the industry forefront.
MAIKONG Competitive Advantages
- 11+ years manufacturing experience
- Multiple international certifications
- Dedicated Nigerian market support
- Comprehensive distributor training programs
- Regular software updates and improvements
- Transparent quality documentation
- Competitive distributor pricing
- Strong after-sales support infrastructure

Technical Specifications
Our quantum health analyzer incorporates advanced technology. The device generates 54 comprehensive health reports. It supports multiple language versions including English. Windows compatibility extends from XP through Windows 11. This ensures broad usability across Nigerian market segments.
| Specification | Details |
| Product Name | Quantum Resonance Magnetic Analyzer |
| Brand | MAIKONG |
| Classification | Class I Medical Device |
| Weight | 3.0 kg |
| Dimensions | 30 x 18.5 x 8.5 cm |
| Reports Generated | 54 detailed health reports |
| Language Support | English, Spanish, French, German, Portuguese, and more |
| Operating System | Windows XP, 7, Vista, 8, 10, 11 |
| Certifications | CE, ISO 13485, ISO 9001 |
| Warranty Period | 1 Year Comprehensive |
| Report Display | Web and PDF formats |
| Testing Time | Approximately 2 minutes |
Making an Informed Decision on Your Quantum Health Analyzer Purchase

Selecting the right quantum health analyzer requires careful consideration. Certifications provide objective quality indicators. They remove guesswork from the evaluation process. Yet certifications alone don’t guarantee success. Understanding your specific needs matters equally.
Consider your target market and application environment. Clinic use demands different features than home wellness consultations. Patient volume affects durability requirements. Language support matters in multilingual environments. MAIKONG devices offer flexibility across these variables.
Key Questions Before Purchase
Does the device carry current CE and ISO certifications?
Verify that certification dates remain current. Expired certificates indicate lapsed quality systems. MAIKONG maintains active certifications with regular renewal cycles. We provide updated documentation upon request.
What warranty and support comes included?
Understand exactly what warranty coverage includes. Clarify response times for technical support. MAIKONG offers comprehensive one-year warranty with online technical support specifically for Nigerian customers.
Is the software compatible with my existing computer systems?
Check operating system requirements carefully. MAIKONG supports Windows XP through Windows 11. We do not support Android, Mac OS, Linux, or Unix systems. Verify your computer meets minimum specifications.
What language options are available for reports?
Language support affects usability in your market. MAIKONG software includes English and multiple other languages. Report generation occurs in your selected language preference.
Can I verify device authenticity before shipment?
Reputable suppliers facilitate authentication. MAIKONG provides serial number verification before shipping. We confirm genuineness of every unit sold through our authorized channels.
What training and documentation comes with purchase?
Comprehensive training materials ensure successful implementation. MAIKONG provides detailed user manuals, video tutorials, and ongoing technical support. Nigerian distributors receive additional business development training.
Evaluating Total Cost of Ownership
Initial purchase price represents only one cost factor. Consider long-term expenses throughout device lifecycle. Maintenance costs and consumable requirements affect profitability. Software update fees may apply with some manufacturers. Training and support costs impact total investment.
MAIKONG offers transparent pricing with no hidden fees. Our one-year warranty covers most maintenance needs. Software updates remain free during warranty period. This predictable cost structure helps business planning. Nigerian distributors appreciate our straightforward pricing model.
- Initial device purchase cost
- Shipping and customs clearance fees
- Training and setup assistance
- Ongoing software updates (free during warranty)
- Consumable replacement items (minimal with MAIKONG)
- Technical support costs (included with purchase)
- Potential revenue generation from device use
Becoming a MAIKONG Quantum Health Analyzer Distributor in Nigeria

MAIKONG actively seeks qualified distributors throughout Nigeria. We believe in partnership rather than transactional relationships. Our distributor program provides comprehensive support for business success. Territory protection ensures your investment remains secure.
Successful distributors understand both health care and business development. They build relationships with clinics and wellness professionals. Marketing and customer service skills prove essential. MAIKONG provides training and resources to develop these capabilities.
Distributor Program Benefits
Our distributor program offers multiple advantages over simple reselling. Authorized distributors receive preferential pricing structures. They access exclusive marketing materials and training resources. Technical support prioritizes distributor inquiries. We share leads from direct customer inquiries in their territory.
Financial Benefits
- Competitive wholesale pricing
- Volume discount incentives
- Flexible payment terms
- High profit margin potential
- Exclusive territory protection
Marketing Support
- Professional marketing materials
- Product demonstration units
- Digital marketing resources
- Trade show support
- Co-branded promotional items
Operational Support
- Comprehensive training programs
- Priority technical support
- Customer lead sharing
- Business development guidance
- Regular product updates
Distributor Requirements
MAIKONG seeks committed partners with growth mindsets. Ideal distributors have existing health care industry connections. Business registration and financial stability matter. We prioritize partners who share our quality-first philosophy. Minimum initial orders ensure serious commitment.

- Registered business entity in Nigeria
- Relevant health care or medical equipment experience
- Financial capacity for initial inventory investment
- Commitment to quality and customer service
- Marketing and sales capabilities
- Willingness to complete MAIKONG training program
- Minimum initial order of 5 units
Getting Started Process
Becoming a MAIKONG distributor follows a straightforward process. Initial contact establishes mutual interest and qualification. We conduct preliminary discussions about territory and business plans. Qualified candidates receive detailed distributor agreements for review. Upon agreement, initial training and order placement proceed.
- Initial contact via WhatsApp or email expressing interest
- Preliminary qualification discussion with MAIKONG team
- Business plan and territory discussion
- Distributor agreement review and execution
- Initial order placement and payment
- Comprehensive training program completion
- Product shipment and customs clearance assistance
- Ongoing support and business development
The entire process typically completes within 2-4 weeks. This timeline depends on documentation speed and customs clearance. MAIKONG guides you through each step. We want our distributors to succeed from day one.
Start Your MAIKONG Distributor Journey Today
Join the growing network of MAIKONG distributors across Nigeria. Access certified quantum health analyzer products, comprehensive training, and dedicated support for your business success.
Conclusion: Investing in Quality Through Proper Certification

MAIKONG Certifications to Look for When Purchasing a Quantum Health Analyzer
The Nigerian health care market offers tremendous opportunity. Quality diagnostic equipment remains in high demand. The quantum health analyzer provides valuable wellness screening capabilities. Yet success depends on selecting properly certified devices.
Certifications protect multiple stakeholders. They ensure patient safety through rigorous testing standards. They safeguard practitioners against liability exposure. They verify manufacturer commitment to quality excellence. Nigerian entrepreneurs benefit from global quality standards.
MAIKONG stands ready to support your health care equipment needs. Our certified quantum health analyzer devices meet international standards. We provide transparent certification documentation and robust support. Our distributor program offers partnership for long-term success.
Don’t compromise on quality in pursuit of lower prices. Certified equipment delivers better patient outcomes and business sustainability. The small premium for proper certification pays dividends through reliability and reputation. Your patients and your business deserve nothing less.
Contact MAIKONG today to discuss your quantum health analyzer needs. Whether purchasing a single unit or establishing a distribution business, we provide expert guidance. Our team understands the Nigerian market and stands ready to support your success.
Take Action Now – Contact MAIKONG
Ready to purchase certified quantum health analyzer equipment or explore distributor opportunities? Our Nigerian support team responds quickly to all inquiries. Multiple contact channels ensure you reach us conveniently.
Website: quantumanalyzer.ng
Order Now: Place Your Order
MAIKONG – Your trusted partner for certified quantum health analyzer devices in Nigeria. Quality you can trust. Support you can rely on. Partnership for success.

